11-08-04 22:14
No 540477
Well i extracted the P2P and i have to say that the yield is not as bad as exspected ~ 24 ml P2P. And I used 63 ml Benzylchlorid and little more than 38 ml Acetonitril. But my P2P is really dark ! dark brown colored , with a really lets call it unpleasant smell , ( little bit like some sort of plant but this must be an ugly one ...)
(The Archetypical "Good Guy")
11-08-04 22:27
No 540479
How did you extract it and verify that it is P2P?
In my experience with this method, you get something from the distillation, with a b.p. not too far for that of P2P (cant remember the exact b.p., lost the notes). It is however NOT p2p...
Regards
Bandil
Only one way leads to Valhalla - Running amok with TOTAL VIKING POWER
(Stranger)
11-08-04 23:34
No 540497
Well started with acidifation of this funny white salt followed by 3 times extraction with 35 ml Et2O( NaCl has been added to mixture .) to remove the yellow color from the bottom (water ) Layer. Et2O extracts pooled together and washed with Water .
Water removed by Dropping funnel and heatng to 110 C. Well ill make a sample with anilin acrylation and compare Products by chromatography. by the way ( wich solvent dont know the english word for it // Fließmittel // works best ?)
And i also looked at the pics by xicori (Meerwein acrylisation...) and his crude >Keton looks exactly like mine !! the only Problem is my Vakuumpump broke . ( u can still turn it on but it makes really unpleasant sounds like sth is scraping insinde ...) Can i destill it without too much loss @ atmospheric pressure ?
(Hive Addict)
11-09-04 17:11
No 540660
The problem isn't excactly the reactivity of the nitrile. One major problem is the fact that the nitrile is acidic enough to protonate the Grignard reagent, turning your precious benzylmagnesium chloride to plain toluene. The deprotonated acetonitrile then goes on and forms different side reaction products. I have a reference to back this up somewhere. Of course one can argue that it is about reactivity because if the reaction with the nitrile was much faster than a proton transfer, this problem would not exist. But proton transfers are known to be very fast reactions.
I think that another problem is the addition of a second molecule of the Grignard reagent to the formed salt of the imine (iminate?), the reason why Grignard reagent should be added to an excess of the nitrile and not the other way around.
However, the reaction is proven to give P2P in literature several times, yields are the problem.
fear fear hate hate
(Hive Bee)
11-09-04 19:30
No 540678
If the problem is deprotonation of acetonitril, then the exact same reaction but instead of hydrolysis with acid, reduction with NaBH4 to amphetamine is consulted should not give an 80% yield1. Am I wrong in my reasoning? As the reduction step comes post-imin formation...
1 JACS 109, 3378-3387 (1987) Procedure adapted from the synthesis of #49 (../rhodium /amphet
(Hive Addict)
11-12-04 19:45
No 541265
Thanks for asking, an interesting example which I had overlooked, even though I've seen it several times before. That preparation is obviously tried and true. I'm convinced that steric factors play a major role here as phenylacetonitrile is more acidic than acetonitrile, and methylmagnesium iodide more basic than benzylmagnesium chloride. Also note the inverted addition. So, I must take my word back about the reactivity of the nitrile group not being the issue. Here is another example of a Grignard addition to nitrile, propionitrile in this case: Post 475862 (moo: "one pot 1-phenyl-2-butanamine etc", Novel Discourse).
Here is the reaction scheme from the original article JACS 109 3378-3387:
fear fear hate hate
(Chief Bee)
11-13-04 01:27
No 541306
(Rated as: excellent)
Novel diacid accelerated borane reducing agent for imines
Zhi-Hui Lu, Nandkumar Bhongle, Xiping Su, Seth Ribe and Chris H. Senanayake
Tetrahedron Letters 43, 8617–8620 (2002) (../rhodium/pdf /grignardimin
Abstract
A remarkable effect of diacids in modulating the reactivity of borane has been discovered. This novel process provides a rapid and excellent access for reduction of a variety of imines with different functionalities.
____ ___ __ _
New resolution approach for large-scale preparation of enantiopure didesmethylsibutramine (DDMS)
Zhengxu Han, Dhileepkumar Krishnamurthy, Q. Kevin Fang, Stephen A. Wald and Chris H. Senanayake
Tetrahedron: Asymmetry 14, 3553–3556 (2003) (../rhodium/pdf /grignardimin
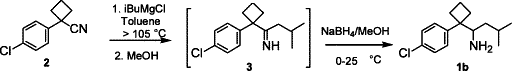
Abstract
An improved synthesis and efficient resolution method to prepare both enantiopures of DDMS using crystallization of enantiomerically pure tartaric acid salts of racemic DDMS are disclosed.
Experimental
A 1 L three-necked round-bottomed flask was charged with 1-(4-chlorophenyl)-1-cyclobutylcarbonitr
____ ___ __ _
A Study and Identification of Potential By-Products of Sibutramine
G. Om Reddy, M. R. Sarma, B. Chandrasekhar, J. Moses Babu, A. S. R. Prasad, and C. M. Haricharan Raju
Organic Process Research & Development 3, 488-492 (1999) (../rhodium/pdf /grignardimin
Abstract
In the synthesis and process development of sibutramine (9), the isolation and characterization of two potential by-products namely heptane dinitriles (4a-b) and bis-cyclobutyl alkylamine (10) have been studied. The key steps in the synthesis of sibutramine which have contributed to the formation of above by-products are cycloalkylation of 4-chlorophenyl acetonitrile (1) and tandem Grignard reduction on 1-(4-chlorophenyl)-cyclobutyl carbonitrile (3).
____ ___ __ _
Synthesis of sibutramine, a novel cyclobutylalkylamine useful in the treatment of obesity, and its major human metabolites
J.E. Jeffery, F.Kerrigan, T.K. Miller, G.J. Smith and G.B. Tometzki, J. Chem. Soc. Perkin Trans. 1, 2583-2589 (1996)
DOI:10.1039/P19960002583
Abstract
Synthetic routes to N-{1-[1-(4-chlorophenyl)cyclobutyl]-3-me
The Hive - Clandestine Chemists Without Borders