06-04-04 03:39
No 511411
i saw on rodium that catechol is turned into methyenedioxy benzene with CH2Br2..
can a same kind of thing be done gallic acid?ie.
use CH3Br on the sodium salt of gallic acid?
(Stranger)
06-04-04 04:06
No 511418
to make what product? 4-hydroxy-1,3-benzodioxole-5-carboxylic acid?
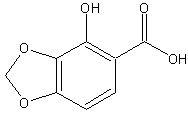
I think it would, but would the 3,4 or 4,5 configuration be favoured?
(Hive Bee)
06-04-04 04:22
No 511426
carbocation: you confused 2,3,4-trihydroxy-benzoic acid for galic acid (= 3,4,5-trihydroxy-benzoic acid)
pink_dust_angel: you confused methylenation (forming -O-CH2-O- on catecholes) with methylation (forming -O-CH3 on phenoles) which would yield 3,4,5-trimethoxy-benzoic acid
�The real drug-problem is that we need more and better drugs.� � J. Ott
(Stranger)
06-04-04 05:20
No 511432
if u actually read my post properly u would realize i was describing a different chemical.....
i said could you use CH3Br (NOT CH2Br2) to turn the OH groups on the bezene of gallic acid into Methyl ethers ie. O-CH3
this is the reaction(look in ure stolen, school library org. chem book):
HBr + MeOH --> MeBr + H2O
MeBr + R-O(-)Na(+) --> Me-O-R + BrNa
sorry to patronise by showng u that lil reaction i just wanted to make sure u knew what i was talkin about
(Chief Bee)
06-04-04 10:45
No 511468
../rhodium /mescali
The Hive - Clandestine Chemists Without Borders
(Stranger)
06-05-04 03:36
No 511604
ok thnx for that..i read it b4 which is what gave me the idea for this
just out of interest though:
i was just wandering if the OH group could be made to an ether "old school" style with an alkyl chloride.
ie
gallicacid 4Na(+) + 4CH3Br -->
345-trimethoxy-Benyl-methylester + 4NaBr
(Chief Bee)
06-05-04 06:15
No 511609
i was just wandering if the OH group could be made to an ether "old school" style with an alkyl chloride.
Sure it can, but methyl bromide is seldom used, as it is a gas at reasonable temperatures and hence reactions involving it needs to be performed under pressure. Methyl iodide is therefore the only methyl halide which is of practical use (and because it is more expensive than (CH3)2SO4 the latter is almost universally used, as the methylation of gallic acid to methyl 3,4,5-trimethoxybenzoate consumes four equivalents of the methylation agent).
The Hive - Clandestine Chemists Without Borders