02-27-02 06:22
No 274288
Here is a really interesting idea:
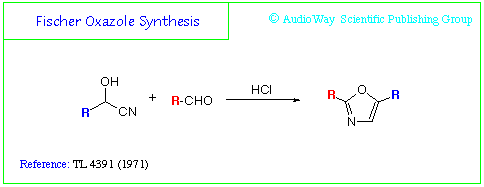
If:
R = Phenyl/C6H5-
R = Amine/NH2-
Then:
RC(OH)CN = Mandelonitrile
RCHO = Formamide
And the product = Aminorex
Synthesis of Mandelonitrile From Benzaldehyde
It seems very very easy to me. See:
../rhodium /mandeli
Preparation of Formamide
Titrate a solution of aqueous ammonia with formic acid of any reasonable strength, and evaporate the water, followed by distillation. (Just boil the water away, keep heating to dehydrate the ammonium formate and vaporize the formamide, and collect the condensate in a recieving flask.)
Now correct me if I'm wrong, but I don't recall this being discussed before. You know, it's kind of cool if you think about it. If one used calcium hypochlorite to oxidize ethanol to ethanal and trichlorinate it, while the resulting calcium hydroxide induces C-C fission, one will form calcium formate and chloroform. Liberation of formic acid for distillation is possible with hydrochloric acid.
Ah, and yes, that lovely byproduct, chloroform.
Don't have access to benzaldehyde? I've always wanted to see the writeups for the chlorination of toluene with.......you guessed it, more calcium hypochlorite. Monochlorination to benzyl chloride is well known, but the patent also says it can be used in excess to procure benzal chloride, which of course reacts with sodium hydroxide to form a gem diol, that spontaneously dehydrates to yield benzaldehyde.
That's pretty fucking cool for an over the counter synthesis, if I do say so myself. What do you guys think about this one? Give me feedback, I got a lot more coming in the near future that I've been thinking about.
PrimoPyro
The Water Will Be Your Only Mirror
(Distinctive Doe)
02-27-02 07:12
No 274301
Good Idea
But there are some health problems/risks with Aminorex.
I don't think I would take aminorex, but methylenedioxyaminorex might bee worth trying.
Here is heath info, for anyone who considers making this stuff.
http://thoracic.org/ic/ic97/tues1.html
http://www.netlaw.net/europe_diet.html
http://www.nhlbi.nih.gov/health/public/l
Read that NIH link about Primary Pulmonary Hypertension, it is definately something you DON'T want. This is the same problem that occured with Fenfluramine.
Thought You Should Know
Foxy
Make a Difference Donate Now!! (http://www.lp.org/drugwar/)
(Distinctive Doe)
02-27-02 07:59
No 274308
Ok Back to the subject
If anyone can translate German, in this patent they make aminorex with styrene oxide(styrene epoxide) with guanidine.
Patent DE2101424
Hmmm it makes me wonder
Could you use beta-Methylstyrene epoxide and get 4-methylaminorex?
Seems like it to Foxy
If so, then isosafrole epoxide would give you the methylenedioxy analog of 4-methylaminorex.
Now that would bee really interesting, and the synth looks fairly easy.
Patent US4311840 has several references for the production of 4-methylaminorex by two different methods. The old CNBr and the urea method which I think Rhodium posted on the Hive earlier.
Make a Difference Donate Now!! (http://www.lp.org/drugwar/)
(Hive Prodigy)
02-27-02 08:16
No 274313
Now that is an even better idea!
Piperonal would be the starting point for the mandelonitrile derivative of course, and many of us are already familiar with such a compound.
To bee honest, I like your reaction better, I just liked this one because it was a completely unthought of idea to me, and formamide is so easy to make compared to other reagents like CNBr....
PrimoPyro
The Water Will Be Your Only Mirror
(Hive Bee)
02-27-02 08:27
No 274319
Example 1
---------
A mixture consisting of 120g styrene epoxide, 65g guanidine freebase and
800ml abs. EtOH is refluxed for 4h, the solvent distilled off and the
remainding oil disolved in 10% HCl, extracted 3 times with ether, the
aqueous phase made strongly alcaline with 50% NaOH, the secreted oil
dissolved in 400ml EtOH and the so obtained solution saturated with CO2.
One obtains 95g of the hydrogencarbonate of
beta-hydroxy-beta-phenylethylguanidine. F.143-4�
{here is a stamp "bad original" - is there something missing??}
Example 2
---------
A mixture of 11.0g styrene epoxide and 21.1g N,N-diphenylguanidine in
200ml abs. EtOH is refluxed for 6h, the solvent distilled off and the
residue treated with 2N HCl, filtered, extracted with Et2O, the aqueous
phase made alcaline with 50% NaOH, extracted with ether, the etheric
layer dried over anhydr. K2CO3, evaporated to dryness and the residue
recrystalised from benzene. one gets the 2-amino-5-phenyl-2-oxazoline,
with the same properties as the product obtained in example 1.
Example 3
---------
A mixture of 22.0g styrene epoxide and 42.3g N,N-diphenylguanidine in
250ml IPA is refluxed for 3h, the solvent removed under vacuum, the
residue treated with 2N HCl and worked up as in example 2. one obtains
thus 2-amino-5-phenyl-2-oxazolin which is identical with the product
obtained in example 1.
Claims
------
1. process for the production of 2-amino-5-phenyl-2-oxazolin, where one
reacts styrene epoxide with a substance of the general formula 1 where
R1 is an aminogroup or an aminogroup substituted with 2 phenyls, R2 an
iminogroup, or R1 and R2 mean a single nitrogen atom, where in the case
that R1 is an aminogroup and R2 an iminogroup one obtains as
intermediate product beta-hydroxy-beta-phenylethylguanidine, which
cyclisizes with elimination of NH3.
2. process like claim 1, where cyclisation is done at high temperature and
preferably low pressure.
3. process like claim 1, where the reaction is done in an organic
solvent, preferably in a low alcohol.
tell me if you need the main part of the patent!
(Hive Bee)
02-27-02 08:33
No 274322
what do you think of this:
(substituted) propenyl benzene ---> pseudonitrosite ---(1.AA, 2.reduction of nitro group, 3.hydrolysis)---> (substituted!) phenylpropanolamine and then the cyanate method of other thread.
???
(Hive Prodigy)
02-27-02 08:35
No 274323
I think it would be easier to use N-bromosuccinimide with water to form the alpha-hydroxy-beta-bromo (substituted)phenylpropane, followed by reaction with ammonia to form PPA.
PrimoPyro
The Water Will Be Your Only Mirror
(Chief Bee)
02-27-02 12:05
No 274394
Primopyro: The designation R-CHO in the diagram at the top signifies an aldehyde, and I am not sure if formamide qualifies as one. Formic acid is an aldehyde and an acid at the same time (H-COOH or HO-CHO) but it still does not react as an aldehyde.
Foxy: Starting with a propenylbenzene (and then making the epoxide with either oxone or CH3CN/H2O2) would give a suitable epoxide precursor for 4-MAR. If a chiral auxillary is used in the epoxidation step (featured in many articles), you will be able to get exclusively the E- or Z-epoxide, so that you can control the stereochemistry of the 4-MAR product (cis or trans). The cis and trans forms will still be racemic though, as the nucleophilic attack by guanidine is only regioselective, not stereoselective.
Great find with those epoxide refs!
(Her Majesty, Stoni's Kitty)
02-27-02 15:17
No 274498
I had a CA reference for that guanidine/styrene epoxide synthesis for aminorex for quite some time now. I was going to include it in Rhodium's previous thread on the subject, but decided to do some research into the methodology before posting anything.
The literature does indeed support the idea that reacting guanidine with ANY epoxide, followed by thermal cleavage of the intermediate, should result in the expected aminorex compound. I have a review on the synthesis of such compounds. I'll have to look for it.
Anyway, I've given some thought as to how one would go about getting intermediates from the 3,4-isoalkene. Oxone seems to be the best OTC way. Next one could either go the guanidine route directly or reaction with hexamine could yield a decent amount of the amino alcohol. OR, the epoxide could be reacted with AlI3 formed in situ in any suitable solvent from I2 (gotten from NaI or KI and oxone) and Al to form the halohydrin. This could then be reacted with an amine or azide or whatever to eventually get the desired amino alcohol. These methods are the most OTC that I can think of.
As for the guanidine issue, finding calcium cyanamide has been most difficult. I was really on a roll with the guanidine synthesis until I found out that calcium cyanamide is not really available anymore (except to treat alcoholism, I guess). As an agricultural product, it seems to have become obsolete. But the guanidine synthesis from calcium cyanamide does seem to still bee in many of the newer patents as the preferred route.
Foxy 2, you should look for that paper describing the CNS effects of various aminorex analogues. I gave all of the relevant information in Rhodium's last thread on the subject. I tried a basic volume by volume search in CA about four years ago using the names of the authors as keywords but found nothing. YOU however, seem to have a real knack for using online sources. If anyone can find that paper it's you, FOXY2 . . . Go get it you gorgeous distinctive doe!