(Distinctive Doe)
03-24-02 22:57
No 287381
(Rated as: excellent)
Here is a new one, well its an old one but new to the Hive.
Its an analog of pemoline.
Other names: Fenozolone; Phenozolone; LD 3394;
2-Oxazolin-4-one, Ordinator;
2-(Ethylamino)-4-oxo-5-phenyl-D2-oxazoli
Appetite depressant 2-ethylamino-5-phenyloxazolin-4-one.
(Laboratoires Dausse S. A.). Ger. (1969)
DE 1297108 (6-12-1969)
Abstract
The title compd. (I) is prepd. by cyclization of N-ethyl-N'-(a- phenyl-a-chloroacetyl)urea (II) in the presence of NaOEt as an HCl acceptor. II itself is prepd. from EtNHCONH2 and PhCHClCOCl. Thus, a stirred suspension of 17.6 g. EtNHCONH2 in 150 cc. dry C6H6 was treated dropwise (20 min.) with a soln. of 18.9 g. PhCHClCOCl in 100 cc. C6H6, the mixt. kept 15 min. at room temp., then refluxed 5 hrs., and the solvent evapd. to yield 19.6 g. II, m. 146° (C6H6). A suspension of 165.8 g. II in 900 cc. abs. EtOH was treated with a soln. of 15.8 g. Na in 900 cc. EtOH, the mixt. refluxed 2 hrs., kept overnight at room temp., the ppt. sepd., the filtrate evapd., and the residue triturated with ice-H2O to yield 87% I, m. 148° (C6H6). I is an appetite depressant, and has a higher therapeutic index than the 2-methylamino analog.
Here is something else interesting.
The stimulating activity of three higher homologs of 5-phenyl-2-amino-4-oxazolinone [on the central nervous system].
Giudicelli, Rene; Najer, Henri; Prouteau, Monique; Sarret, Monique.
Compt. Rend. (1962), 254 2862-4.
Journal written in Unavailable.
CAN 57:11186 AN 1962:411186
Abstract
Substitution of a straight-chain C1-3 alkyl group on the 2-amino group of 5-phenyl-2-amino-4-oxazolinone increases its central exciting properties.
So that means these are more centrally exciteing than pemoline! Worthwhile, who knows... maybee
(Distinctive Doe)
03-24-02 23:13
No 287388
(Rated as: excellent)
5-phenyl-2-ethylamino-4-oxazolinone and its preparation
Patent GB963375
Abstract
----------------------------------------
The invention comprises 5-phenyl -2- ethylamino -4- oxazolinone, its preparation by heating an N1-ethyl-N11-(a -phenyl -a - haloacetyl) urea in the presence of a hydrogen halide binding agent, and pharmaceutical preparations containing it. The starting materials are prepared by condensing N-ethylurea with an a -phenyl -a - haloacetyl halide. The compound of the invention may be used as a psychotonic, an "anti-fatigue" agent or an anorexigenic, and may be administered in the form of pharmaceutical preparations (e.g. tablets) containing it together with a diluent.
A new prepn. for 5-phenyl-2-methylamino-4-oxazolinone (I) was developed, and 7 new similar compds. were prepd. EtONa, made from 3.75 g. Na in 250 ml. EtOH, was added to a suspension of 37 g. 1-methyl-3-(a-phenyl-a-chloroacetyl)urea
Those who give up essential liberties for temporary safety deserve neither liberty nor safety
(Distinctive Doe)
03-24-02 23:44
No 287394
(Rated as: excellent)
Synthesis of some 2-imino-4-oxazolidinones. VI. 5-Phenyl-2-cycloalkylamino-4-oxazolinone
Najer, Henry; Giudieelli, Rene; Menin, Jacques.
Bull. Soc. Chim. France (1963), (8-9), 1810-13.
Abstract
cf. CA 59, 589b. PhCHClCOCl (I) condensed with a cycloalkylurea gave N-cycloalkyl-N°-(a-phenyl-a-chloroacetyl
5-Aryl-2-cycloalkylamino-4-oxazolinones.
Patent NL6613484 (It has the deatails)
Abstract
The title compds. of the formula I (R = Ph or p-C6H4Cl and R1 = H or Me) were prepd. by treating p-ClC6H4CHClCO2Cl (II) or PhCHClCO2Cl (III) with cyclopropylurea (IV) or N-cyclopropyl-N-methylurea (V) and treating the product (VI) with Na alcoholate. I are useful in prepg. psychostimulant, anorexigenic, or sedative compns. Thus, 39 g. II in 130 cc. C6H6 was added through a dropping funnel over 45 min. to a mixt. of 17.6 g. IV, 21.3 g. PhNMe2, and 310 ml. C6H6, after 1 hr., the mixt. refluxed 5 hrs., the oily residue poured out, the C6H6 distd., the residue washed 3 times in 100 ml. Et2O, and the Et2O evapd. in vacuo to yield 28.6 g. N-cyclopropyl-N'-[a-(p-chlorophenyl)-a-c
Tautomerism of 5-phenyl-2-alkyl(or aralkyl)imino-4- oxazolidones with 5-phenyl-2-alkyl(or aralkyl)amino-4-oxazolinones.
Najer, Henry; Giudicelli, Rene; Menin, Jacques; Loiseau, Jacques.
Compt. Rend. (1962), 254 2173-5.
Abstract
cf. CA 55, 27268i. Refluxing PhClCHCOCl over (MeNH)2CO in C6H6, with Et3N present, gave H. Heating Me2NH with 2-imino-5-phenyl-4-oxazolidinone in EtOH in a sealed tube at 120° gave III. Unexpectedly, III proved to be a central nervous system stimulant, as were I but II was not. Comparison of the ultraviolet and infrared spectra confirmed the suspicion that I exist predominantly in the tautomeric aminooxazolinone (IV) forms. In alcohol, the l and log e, resp., are: II, 209, 4.24; III, 228, 4.43. The series IV: (R .dbd. Me), 221, 4.48; (R .dbd. Et), 221, 4.42; (R .dbd. Pr), 222, 4.46; (R .dbd. iso-Pr), 221, 4.44; (R .dbd. Bu), 223, 4.34; (R .dbd. Bz), 225, 4.48; (R .dbd. phenylisopropyl), 225 mm, 4.46. The infrared comparisons, limited to the bands of valence vibrations of CO and C: N bonds, assign conjugated double bonds to the tautomers for which these frequencies are lowered (Bellamy, The Infrared Spectra of Complex Molecules, 1954 (CA 48, 12562c)). Found for CO, C:N, resp.: II, 1765, 1700; III, 1730, 1630 cm.-1. The same decrease (CO, 1720-1745 and C:N, 1645-1680 cm.-1) is noted on comparison of the infrared spectra of IV with that of II.
Those who give up essential liberties for temporary safety deserve neither liberty nor safety
(Distinctive Doe)
03-24-02 23:49
No 287395
(Rated as: excellent)
5-Phenyl-2-cycloalkylamino-4-oxazolinone
Patent GB1005738
Abstract
Ureas of the general formula PhCHX-CONHCONHR (I), where X is a halogen and R is a C3-6 cycloalkyl group, are cyclized in the presence of a Na alkoxide to give the title compds. Thus, 33.3 g. PhCHClCOCl in 130 ml. anhyd. C6H6 is added in 45 min. to a mixt. of 17.6 g. cyclopropylurea and 21.3 g. PhNMe2 in 310 ml. anhyd. C6H6 and the mixt. kept 1 hr. and refluxed 5 hrs. to give 55% N-cyclopropyl-N1-(a-phenyl-a-chloroacety
Pharmacological properties of two derivatives of 5-phenyl-2-cyclopropylamino-4-oxazolinon
Giudicelli, Rene; Najer, Henry; Menin, Jacques; Prouteau, Monique.
C. R. Hebd. Seances Acad. Sci., Ser. D (1967), 265(2), 165-8.
Journal in French. CAN 67:73543
Abstract
2-(N-Methyl-N-cyclopropylamino)-5-phenyl
(Distinctive Doe)
03-25-02 03:04
No 287458
(Rated as: excellent)
5-Phenyl-2-cyclopropylamino-4-oxazolinon
I is approximately equal in strength to dl-amphetamine except it has a larger safe therapeutic safety window.
II had equal stimulateing properties to d-amphetamine and is longer lasting. Its is less toxic orally and has only a very slight cardio-vascular effect in the cat or dog in doses up to 5 mg/kg administered parenterally(thats quite high doses).
Who knows what the other risks are for these compounds, mentally and physically. They seem like VERY interesting canidates for experimentation. Several recent patents have mentioned the n-ethyl analog, which leads me to believe that serious toxicity issues have yet to bee found. Wonder what the substituted analogs are like? ie. methylenedioxy, trimethoxy, 4-Br-2,5-dimethoxy ect ect
Who's going to bee the Shulgin of oxazolinone's???
Those who give up essential liberties for temporary safety deserve neither liberty nor safety
(Distinctive Doe)
03-25-02 04:45
No 287487
(Rated as: excellent)
a-Chlorophenylacetyl chloride.
Indian Patent 146686 (8-11-1979)
Abstract
a-Chlorophenylacetyl chloride was prepd. in 72% yield by treatment of mandelic acid with SO2Cl2 (see thread Post 208448 (Osmium: "Re: Making SO2Cl2", Novel Discourse) for prep of SO2Cl2) in the presence of DMF at room temp for 80 h.
Other methods
Patent US4639541
Patent GB1374324
The following two patents go together
Patent US3794679
Patent US3812183
Patent GB1425304
Those who give up essential liberties for temporary safety deserve neither liberty nor safety
(Distinctive Doe)
03-25-02 05:17
No 287498
(Rated as: excellent)
What do ya know, this made the mice happy!!!
And its easy to make from Pemoline(see Rhodiums for the synth of Pemoline). The oral LD/50 for mice is mice and huge for this one(250-1000 mg/kg).
Synthesis and antidepressant activity of 5-phenyl-2-(2-propynylamino)-2-oxazolin-
Lee, Cheuk-Man; Horrom, Bruce W.; Michaels, Raymond J.; Rasmussen, Ronald M.; Plotnikoff, Nicholas P.
J. Med. Chem. (1972), 15(12), 1252-5.
Abstract
Propargylpemoline [5-phenyl-2-(2-propynylamino)-2-oxazolin
2-Propargylamino-5-phenyl-2-oxazolin-4-o
Patent US3578672
Abstract
The title compds. (I, X is, e.g., H, 4-Cl, 2-Me, or 3,4,5-(MeO)3; R is H, Me, or Ph; R1, R2 is H or Me) were prepd. from 2-amino-5-phenyl-2-oxazolin-4-one and propargylamines, or from 1-(chlorophenylacetyl)-3-(1,1-dimethyl-2
Those who give up essential liberties for temporary safety deserve neither liberty nor safety
(Distinctive Doe)
03-25-02 05:37
No 287500
(Rated as: excellent)
2-(Diethylamino)-5-phenyl-2-oxazolin-4-o
Patent DE1237570 (in German)
Abstract
A mixt. of 176.1 g. 2-imino-5-phenyl-2-oxazolin-4-one and 648 g. Ac2O was heated at 100° with stirring to give 87% 2-acetamido-5-phenyl-2-oxazolin-4-one (I), m. 194-6°. Et2NH (219.3 g.) was added in small portions with stirring at room temp. to 218.1 g. I to give 85% the title compd. (II), m. 74-6°.
Compounds producing central nervous system stimulation and anorexia.
Patent US3313688
Abstract
I, where R1 and R2 are lower alkyl, and their acid addn. salts are prepd. Thus, a mixt. of 8.8 g. 5-phenyl-2-imino-4-oxazolidinone, 9 g. dimethylamine, and 50 ml. ethanol is heated in an autoclave at 125° for 2 hrs. and cooled, and the ethanol removed to give 2-dimethylamino-5-phenyl-2-oxazolin-4-on
Dimethyl analog
Patent US3665075
Those who give up essential liberties for temporary safety deserve neither liberty nor safety
(Distinctive Doe)
03-25-02 05:50
No 287503
(Rated as: excellent)
2-Amino-2-oxazolin-4-ones. I. Synthesis.
Howell, Charles F.; Quinones, Nicanor Q.; Hardy, Robert A., JR.
J. Org. Chem. (1962), 27 1079-85.
Abstract
A series of new 5-aryl-2-dialkylamino-2-oxazolin-4-ones has been prepd, by (a) condensation of mandelic acid derivs, with dialkylcyanamides, (b) aminolysis or (c) alkylation of 2-amino-5-aryl-2-oxazolin-4-ones, and (d) aminolysis of 5-aryl-2-thio-2,4-oxazolidinedioneswith secondary amines. Pharmacol. results indicate that 2-dimethylamino-5-phenyl-2-oxazolin-4-on
2-Amino-2-oxazolin-4-ones. II. Tautomerism.
Howell, Charles F.; Quinones, Nicanor Q.; Hardy, Robert A., JR.
J. Org. Chem. (1962), 27 1680-91.
Abstract
Comparison of the phys. and chem. properties of 2-amino-5-phenyl-2-oxazolin-4-one with mono- and dimethyl homologs of known tautomeric structure has shown that the 2-amino-2-oxazolin-4-one structure is indicated rather than the 2-imino-4-oxazolidinone structure previously formulated. The latter form, however, prevails in the conjugated 5-phenyl-2-phenylimino-4-oxazolidinone.
2-Amino-2-oxazolin-4-ones. III. Spectral studies.
Howell, Charles F.; Fulmor, William; Quinones, Nicanor Q.; Hardy, Robert A., Jr.
J. Org. Chem. (1964), 29(2), 370-3.
Central nervous-system stimulant.
The synthesis of 5-phenyl-2-methylimino-4-oxazolidinone.
Patent US3047461
5-Phenyl-2-dialkylamino-2-oxazolin-4-one
Patent US3037990
(Official Hive Translator)
03-25-02 10:15
No 287570
This is quite a work, Foxy! I hope someone, someday, will make use of this information! Not to mention the possible analogues, as well as purely pharmacological implications this comp'd produces.
Let me too throw a dice - for the visually impaired people like myself, here's how it looks:
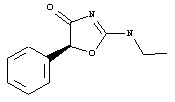
Antoncho
(Distinctive Doe)
03-25-02 10:25
No 287577
Antoncho
The real goodies are the N-cyclopropyl and the N-cyclopropyl-N-methyl. Those two are right up there with amphetamine in strength.
Those who give up essential liberties for temporary safety deserve neither liberty nor safety