07-17-04 14:22
No 519939
Ok this is a long shot but who knows... perhaps someone could interpret this:
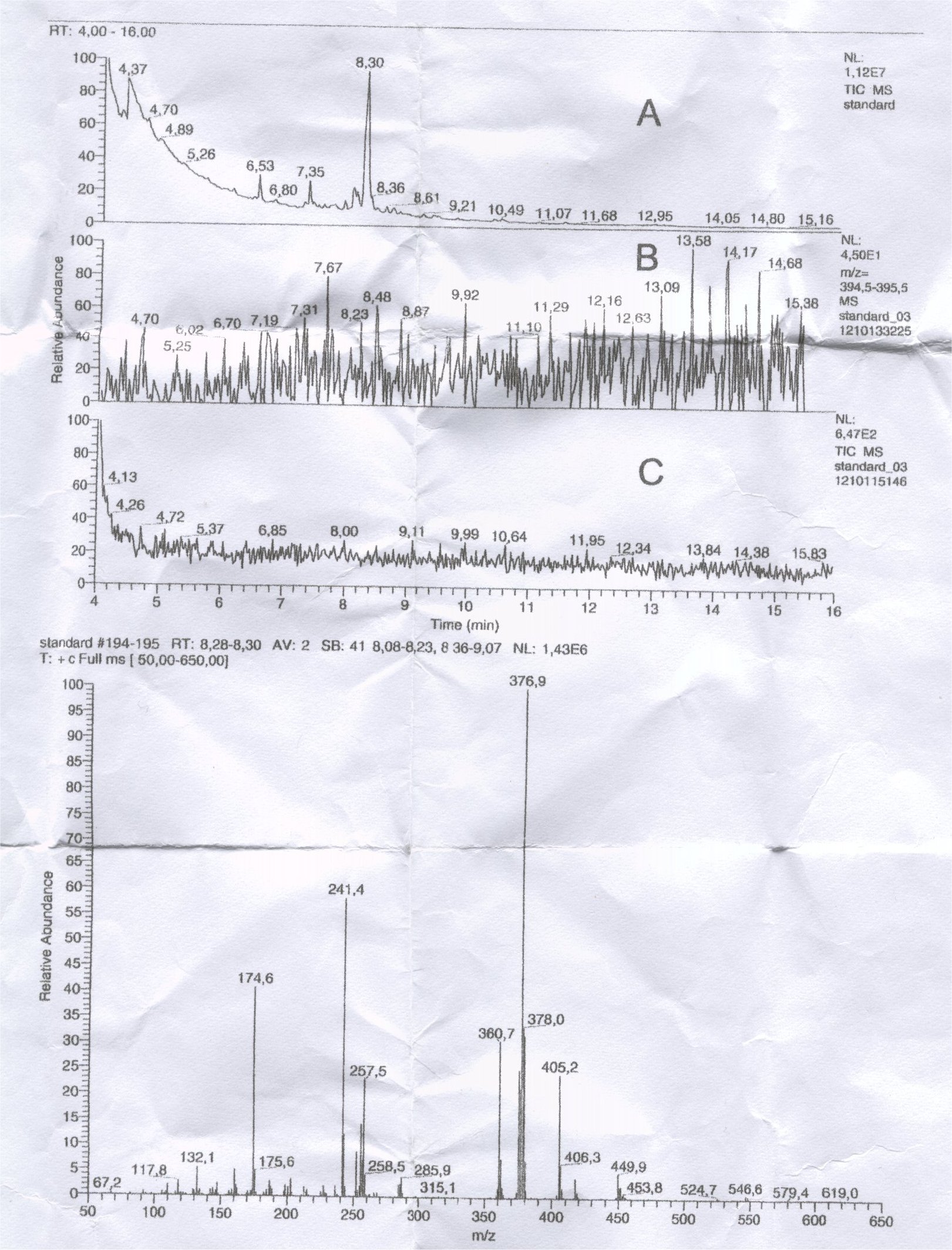
Chromatogram A: positive chemical ionisation with CH4, the most visual peak at 8,30 min
Chromatrogram B: postivie electron impact ionisation of sample (BSTFA+1% TMCIS), selected mass trace m/z 396
Chromatogram C: MS/MS
Van Urk –Salkowski reagent test colors skyblue, the compound is UV fluorescent and it was supposed to be LSD freebase but the chromatograms seems to say otherwise.
Does anybody have suggestions ?
Hippler
(Newbee)
07-17-04 15:43
No 519963
I would like to help you, but are you sure the endproduct you are looking for is LSD ? Chromatogram B is made by tracing m/z 395 so an incorrect mass has been looked at? Which synthetic route was used to produce this so-called LSD freebase ?
A little poison now and then: that makes for agreeable dreams.
(Hive Bee)
07-17-04 15:51
No 519964
The analysis was not performed by me and am mostly puzzled about this aswell.
Highly doubtfull but possible is that the person was looking for the mass of lysergic acid diethylamide dihydrate (mass 359,46).
Hippler
(Newbee)
07-17-04 16:30
No 519974
Hmm, a nice puzzle you gave me Vaaguh....From the CI data it is difficult to tell what the mol mass is of this unknown compound. There is no clear M+1(H)/M+17(CH5) combination as you would expect for a methane CI measurement. The fragmention pattern is unknown to me and not comparable with the LSD (silylated) spectrum I´ve from literature. What else could have been attached to the acid group? You used diethylamine of sufficient purity ?
Damn, there goes my saturday night
A little poison now and then: that makes for agreeable dreams.
(Hive Bee)
07-17-04 16:43
No 519978
You used diethylamine of sufficient purity
Not sure but assumed so ![]() (CDI method by the way), thanks in advance for helping me out here
(CDI method by the way), thanks in advance for helping me out here
Hippler