10-10-04 08:33
No 535158
I got a bottle of formic acid recently, but there's no sign on the label about the purity/concentration of it.
How can I tell more or less accurately with a simple method? And if it happends to be too dilluted, can I just boil it to get to the 85% needed for the performic oxidation of isosafrole?
(Hive Bee)
10-10-04 11:12
No 535166
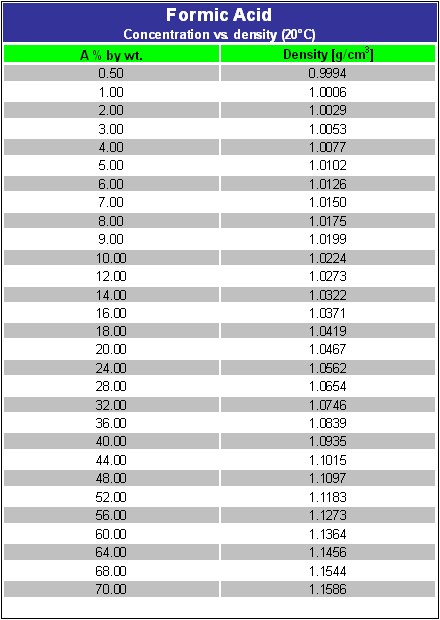
Get yourself an aerometer and mesure the density of the formic acid. That's the easiest and fasted method to measure the concentration of most solvents.
“The real drug-problem is that we need more and better drugs.” – J. Ott
(Balanced Ego)
10-10-04 11:12
No 535167
> How can I tell more or less accurately with a simple method?
titration of a diluted sample - get a burette and a graduated flask.
formic acid is usually titrated against KMnO4, but if you can't get
that, then titration against a strong base will work too.
> can I just boil it to get to the 85%
no - bp of formic acid is 100.7°C! you could have found that out yourself,
lazy bastard!
"And you for sure cant read nor write assembler, idiot." - orgy.
(Chief Bee)
10-11-04 01:00
No 535244
The Hive - Clandestine Chemists Without Borders