Fig. 1.
Assembled apparatus for the generation of HCl(g)
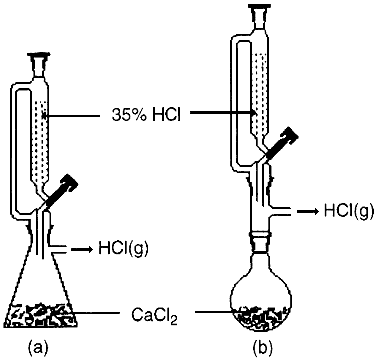
Hydrogen chloride is an important chemical of interest not only by itself but also for preparative purposes. Although available in cylinders of variable capacity and purity, generation of HCl gas is done frequently in chemistry labs.
Hydrogen chloride can bo prepared by a number of methods including1:
- Dehydrochlorination processes
- Reaction of chlorine with hydrogen and hydrogenated compounds
- Hydrolysis and hydrogenolysis of some chlorides
- Protonation of the chloride ion by strong acids and
- Liberation from aqueous solutions
However, most procedures found in preparative handbooks make use of concentrated H2SO42,3, for example:
Eq. 1:
NaCl + H2SO4 = NaHSO4 + HCl(g)
Eq. 2:
HCl·(H2O)x + H2SO4 = H2SO4·(H2O)x + HCl(g)
These procedures are the most universally adopted because they work satisfactorily with quite inexpensive chemicals.
Eq. 1 can be explained in terms of a proton transfer from the strong (in the first ionization) non-volatile sulfuric acid to the weakly basic chloride ion. It works at ambient conditions because HCl is a gas. The combination of acidity, redox properties, low volatility, and cost of H2SO4, makes it the acid of choice. The process is conducted conveniently in standard glassware assemblies such as those in the figure. Concentrated H2SO4 is poured through the funnel, and salt is placed in the flask. Even though the generator is properly installed, losses of H2SO4 through the stopcock may be produced eventually.
In Eq. 2 H2SO4 behaves as a water-scavenging agent leading to a system unable to dissolve efficiently the HCl, so it escapes. The well-known dehydrating ability of H2SO4, together with the characteristics mentioned above, invite its use for this purpose.
However, because dehydration of hydrochloric acid can be achieved by reagents other than H2SO4, and because the handling of 98% H2SO4, in the freshman lab should be reduced as much as possible, especially when a significant amount of acid is involved, I want to recommend CaCl2 for the following reasons:
- CaCl2 is considerably safer to handle than concentrated H2SO4.
- It also is available at low cost, sometimes as a byproduct of the Solvay process.
- The standard assemblies for gas generation shown above are appropriate for the preparation of crude HCl(g).
- It is able to fix enough water (over the 30% of its weight considering it hydrates only to CaCl2·2H2O, which is stable up to 150ḞC) to deliver a significant amount of HCl gas. In a typical run, 100 g of 35% HCl were added over 100 g of granular CaCl2 at room temperature (the thermal effect is not significant), and 15 g of HCl(g) were obtained. This represents over 40% yield, which may be compared with the 80%, yield reported2 for the process represented in Eq. 2; however, it uses approximately a 1:3 weight ratio between H2SO4 and HCl(aq).
- CaCl2 also is easier to dispose of than H2SO4, and less base is required to neutralize the final waste.
An additional advantage of this procedure with respect to those based on H2SO4 concerns educational aspects. By heating at 200ḞC, the resulting waxy mixture (or crystalline mass to which it converts after a few hours), the anhydrous CaCl2, can be regenerated for re-use once a solution of HCl is obtained. In this case, some calcium oxychloride formed can be ignored.
Even though this may be without value from a practical viewpoint, it may be of educational significance because it attracts attention to topics of current interest such as optimizing of processes, recycling of chemicals, and reducing of wastes. To this end, the assembly shown in Fig. 1b is more appropriate than that in Fig. 1a because the resulting mixture is ready to be distilled.