Abstract
Synthesis of (1RS,2SR)-(Ī)-2-amino-1-phenyl-1-propanol (2) by reductive amination of (R)-(-)-1-hydroxy-1-phenyl-2-propanone (1) using ammonia and Raney nickel as a catalyst was investigated. Compound 1 was produced by fermentation of molasses with a strain of yeast and benzaldehyde was added during the course of fermentation.
Introduction
(1RS,2SR)-(Ī)-2-Amino-1-phenyl-1-propanol (compound 2) is a good vasoconstricting agent and clinically used as a nasal decongestant.1 This compound is commercially synthesised by chemical methods from propiophenone by a three-step procedure.2
Different methods for the synthesis of 2 are mentioned in the literature,3,4 although the information available on these processes is mostly confined to the patent literature. To the best of our knowledge, it has not been synthesised so far using Raney nickel as a catalyst from (R)-(-)-1-hydroxy-1-phenyl-2-propanone (compound 1) which is obtained by fermentation.
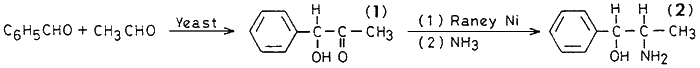
The present paper describes the novel two-step biosynthetic process which involves the production of (R)-(-)-1-hydroxy-1-phenyl-2-propanone by fermentation, which on reductive amination with ammonia using Raney nickel as a catalyst is converted into 2. This two-step process has advantages over the conventional chemical route based on conversion of propiophenone by a three-step synthesis.
Experimental
Production of (R)-(-)-1-hydroxy-1-phenyl-2-propanone (1) by fermentation
Compound 15-11 was produced by fermentation using a strain of yeast which was isolated from rotting apricot. The yeast strain was maintained in yeast-malt-agar slants. The fermentation was carried out in a 100-L fermenter with aeration and agitation. 40 L of the medium, which consisted of molasses (sugar 10%) and ammonium sulphate (0.1%), was charged in the fermenter. It was inoculated with 4 L of 20 hours old seed which was grown separately in a 15-L fermenter (seed and fermentation medium were of identical composition). The fermentation was allowed to proceed for 12 h after which 264 mL of benzaldehyde was added in six divided doses with an interval of 1 h between each dose. The fermentation was continued for 2 days after the addition of the last dose.
Fermentation conditions
The fermenter was agitated at 200 rpm at a temperature of 29Ī1įC and aeration of 11 L/min for a fermentation volume of 44 L.
The fermenter was harvested and the fermented broth passed through a centrifugal separator (Alfa Laval) to remove the yeast. The culture filtrate was extracted with benzene. The benzene extract was concentrated under vacuum below 60įC when crude compound 1 was obtained.
Purification of (R)-(-)-1-hydroxy-1-phenyl-2-propanone (1)
Crude compound 1 (91 g, [α]D25 -100°) was dissolved in 500 mL of ether and shaken quickly with 2x100 mL (5%) of sodium bicarbonate solution for the removal of acids. The combined aqueous phase was extracted with ether and mixed with the supernatant compound 1 solution. The combined ether extract was shaken with 25% aqueous sodium metabisulfate solution (5x100 mL). The aqueous extract was treated with sodium bicarbonate (~300 g) until the evolution of carbon dioxide ceased, and subsequently extracted with ether. Concentration of the extract afforded compound 1 (24 g) which was identified by preparing the following derivatives12.
- 2,4-Dinitrophenylhydrazone: crystallised from ethanol, mp 170Ė171įC (literature cited 170įC).
- Semicarbazone: crystallised from toluene, mp 193Ė194įC (reported 194įC).
Preparation of Raney nickel catalyst
The catalyst was prepared by the method described in the literature.13
Synthesis of (1RS,2SR)-(Ī)-2-amino-1-phenyl-1-propanol (2)
Compound 1 5g [α]D25 -155° (containing 4.28 g pure (R)-(Ė)-1-hydroxy-1-phenyl-2-propanone, 0.03 mol·dm-3) was taken up in 15 mL of benzene. 11.4 mL (0.17 mol·dm-3) of 25% ammonia and 0.5 g Raney nickel were added and hydrogen was passed for 2.30 h at 50 psi. After completion of the reaction, the reaction mixture was filtered off. concentrated hydrochloric acid (7 mL) was added to adjust the pH to 5.5. The solution was concentrated under reduced pressure. The viscous mass was dissolved in absolute ethanol and dry ether was added to the solution. A colourless compound crystallised from absolute ethanol, mp 192Ė194įC (literature cited 190-194įC, [α]D25 0°, was obtained. Yield: 30%, 1.61 g.
Large-scale synthesis of (1RS,2SR)-(Ī)-2-amino-1-phenyl-1-propanol (2)
Compound 1 106 g [α]D25 -122° (containing 71.44 g pure (R)-(Ė)-1-hydroxy-1-phenyl-2-propanone, 0.4 mol·dm-3) was dissolved in 840 mL of benzene; 116 mL of 25% ammonia (1.7 mol·dm-3) and Raney nickel (approximately 4 g) were added and hydrogen was passed for 3 h at 50 psi. The Raney nickel was filtered off, the reaction mixture was concentrated and conc. hydrochloric acid was added to adjust the pH to 5.5. The syrupy mass was dissolved in absolute ethanol and dry ether was added to it. A colourless compound crystallised from absolute ethanol, mp 193-195įC, was obtained, yield: 33%, 29.47 g.
Results and Discussion
Commercial-scale manufacture of this compound is carried out by a three-step synthetic route starting from propiophenone. However, the process we have developed is novel as it involves the production of (R)-(Ė)-1-hydroxy-1-phenyl-2-propanone by fermentation, followed by reductive amination to yield (1RS,2SR)-(Ī)-2-amino-1-phenyl-1-propanol. A two-phase system with benzene or a single-phase system with ethanol were tried for the hydrogenation process. The two-phase system with benzene gave better results. The Raney nickel catalyst can be recycled twice with a 10% make-up. Ammonia was added in excess to avoid synthesis of the dimeric compound. Better yield was obtained when catalytic hydrogenation was carried out at 50 psi. Low-pressure hydrogenation decreased the yield.