11-01-01 22:53
No 231988
These Russians are COOL!!
Indole derivatives. C. Synthesis of w-(3-indolyl)alkyl bromides.
Eryshev, B. Ya.; Dubinin, A. G.; Buyanov, V. N.; Suvorov, N. N.
Khim. Geterotsikl. Soedin. (1974), (11), 1493-5.
CODEN: KGSSAQ Journal written in Russian.
CAN 82:125216 AN 1975:125216
Abstract
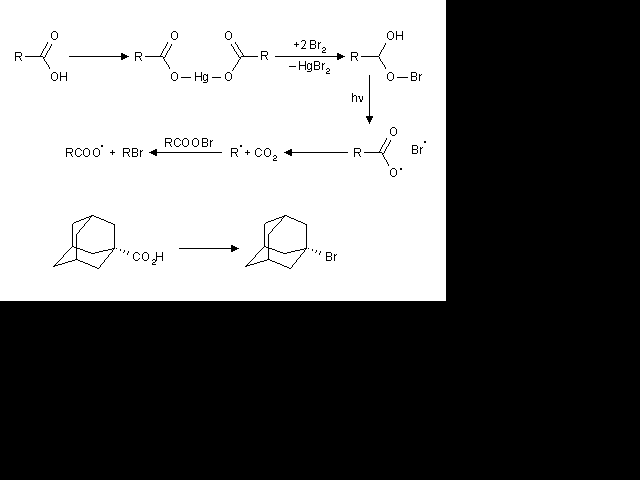
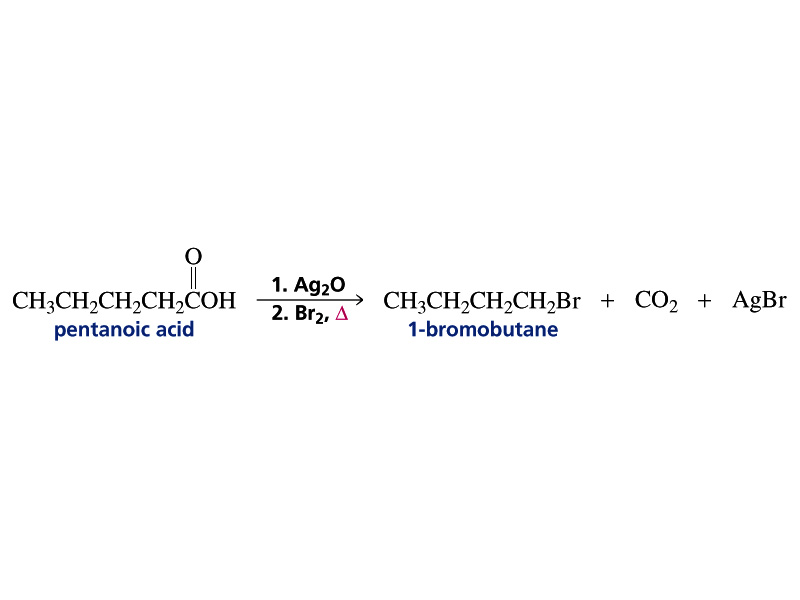
The indolylalkyl bromides I (R = Br; n = 2,3,4,10) were prepd. by Hunsdiecker bromination-decarboxylation
of the Hg salts of I (R = CO2H). Alternately, I (R = Br; n = 3,4) were prepd. by reaction of I (R = HO)
with PBr3 in Et2O.
Hunsdiecker Reaction
Preparation of aklyl halogenides from carboxylic acids.
HgO or Ag2O is used as catalyst. The reaction is via a radical mechanism and must be lightened.
http://fachschaft.cup.uni-muenchen.de/~s
Hunsdiecker Reaction (Borodine Reaction)

C. Hunsdiecker, H. Hunsdiecker and E. Vogt, U.S. pat. 2,176,181 (1939)
H. Hunsdiecker and C Hunsdiecker, Ber. 75, 291 (1942)
A. Borodine, Ann.119, 121 (1861)
R. G. Johnson and R. K. Ingham, Chet. Reus. 56, 219 (1956)
C. V. Wilson, Organic Reactions 9, 341 (1957)
P. I. Abell J. Org. Chem. 22, 769 (1957)
V. Franzen, Chem. Ztg. 81, 138 (1957)
J. W. Wilt and D. D. Oathoudt, J. Ore. Chem. 23 18 (1958)
E. L. Eliel, R. V. Acharya, J. Org. Chem. 24, 151 (1959)
S. J. Cristol and W. C. Firth Jr., J. Org. Chem. 26 280 (1961)
D. H. R. Barton and E. P. Serebryakov, Proc. Chem. Soc. 1962, 309
D. E. Applequist and N. D. Werner, J. Org. Chem. 28, 48 (1963)
F. W. Baker, H. D. Holtz and L. M. Stock, J. Org. Chem. 28, 514 (1963)
J. A. Davis, J. Herynk, S.Carroll, J. Bunds, D. Johnson, J. Org. Chem. 30, 415 (1965)
http://cwx.prenhall.com/bookbind/pubbook
http://cwx.prenhall.com/bookbind/pubbook
Do Your Part To Win The War