(Hive Bee)
09-18-04 00:21
No 531852
(Rated as: excellent)
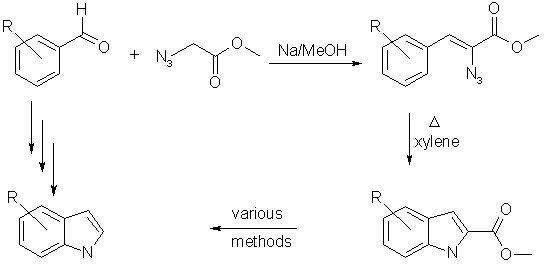
The Hemetsberger-Knittel indol synthesis
With this method it is possible to obtain 4-hydroxyindole/4-benzyloxyindole (ideal precursor for psilocin) and 4-bromoindole (precursor for lysergic acid, see Post 476514 (Lego: "A New Synthesis of Lysergic Acid", Tryptamine Chemistry)
Step 1: Benzaldehydes to alpha-azidocinnamic acid esters
Verbesserte Synthese von alpha-Azidozimtsäure-estern und 2H-Azirinen
Knittel, Dierk
Synthesis, 1985, 2, 186-188

To a solution of sodium (2.3, 0.10 mol) in methanol (50 ml) a mixture of aldehyde 1 (0.05 mol) and methyl azidoacetate 2 (11.5 g, 0.10 mol) is added at a temperature 10-20 °C within 20 minutes. Less soluble aldehydes, e.g. 1f are dissolved in methanol. The mixture is left at room temperatures for 2-3 hours and cooled occasionally to prevent a temperature above 30 °C. The solvent is removed in vacuo at a maximum of 40 °C. After neutralisation of the residue with diluted hydrochloric acid the product is extracted with ethyl acetate. The main part of the less soluble nitro products 3f-h can be obtained by filtration as sparingly soluble crystals. After drying of the extract with calcium chloride and evaporation of the solvent the product is filtered over a column of silica gel (50 g). The column is eluted with petroleum ether (b.p. 70 °C) which contains between 10 (in case of 3a) and 50% benzene (in case of the sparingly soluble products 3f-h). After evaporation of the solvent pure products are obtained which can be further purified by recrystallisation from petroleum ether (b.p. 70 °C) and methanol (Table 1).
Table 1
| Product | R | Yield, % |
| 3a | H | 62 |
| 3b | 4-CH3 | 60 |
| 3c | 4-H3CO | 58 |
| 3d | 4-Cl | 74 |
| 3e | 2,6-di-Cl2 | 55 |
| 3f | 4-NO2 | 55 |
| 3g | 3-NO2 | 80 |
| 3h | 2-NO2 | 6 |
Synthesis of new indolecarboxylic acids related to the plant hormone indoleacetic acid IAA
Flávia A. F. da Rosa, Ricardo A. Rebelo, Maria G. Nascimento
J. Braz. Chem. Soc., 2003, 14(1), 11-15 (http://jbcs.sbq.org.br/jbcs/2003/v14_n1
Abstract: The synthesis of 5,6-methylenedioxy-indol-3-yl-methanoic acid 8 and 5,6-methylenedioxy-indol-3-yl-acetic acid 13 is described. Piperonal was employed as starting material, and the construction of the heterocyclic ring based on the Hemetsberger reaction of the corresponding b-azidostyrene was highly regiospecific. Compound 8 was obtained as a key intermediate towards 13, and a Mannich reaction was used to introduce the required alkyl side chain. The route comprised eight steps giving 13 in 26% overall yield. The formation of the indolic ring via reductive cyclisation of o,b-dinitrostyrene is also presented.
Methyl-2-azido(3,4-methylenedioxyphenyl)
A solution of 3,4-methylenedioxybenzaldehyde (5) (5.0 g, 33 mmol) in methanol (20mL) and methyl azidoacetate (15.3 g, 133 mmol) was added dropwise (1 h) to sodium methoxide solution [prepared from sodium (3.1 g, 135 mmol) in methanol (40 mL)] at –8 °C. The mixture was then stirred for 2 h, maintaining the temperature below 5 °C. The heterogeneous mixture was poured into ice (400 mL) and manually stirred. The yellow suspension was filtered, washed with ice water, and dried in a vacuum oven for 12 h at 70 oC. The yellow solid (7.98 g, 97% yield) was used without further purification in the next step.
Step 2: alpha-azidocinnamic acid esters to indole-2-carboylates
Enazide, 3. Mitt.: Thermolyse von alpha-Azidozimtsestern; Synthese von Indolderivaten
Enazides, III: Thermolysis of alpha-Azido-cinnamtes. Synthesis of Indol Carboxylates
H. Hemetsberger, D. Knittel und H. Weidmann
Monatshefte, 1970, 101, 161-165

In refluxing xylene even after 10 minutes 1 {alpha-azidocinnamic acid esters} are decomposed thermically to yield 90-98% indoles and minor amounts of polymerised products.
| Nr. | R-Azide | R-Indol |
| 5 | --- | --- |
| 6 | p-CH3 | 6-CH3 |
| 7 | o-CH3 | 4-CH3 |
| 8 | p-Cl | 6-Cl |
| 9 | o-Cl | 4-Cl |
| 10 | p-Br | 6-Br |
| 11 | o-Br | 4-Br |
| 12 | p-CH3O | 6-CH3O |
| 13 | m-CH3O | 5- and 7-CH3O |
| 14 | o-CH3O | 4-CH3O |
| 15 | p-F | 6-F |
Abstract: Die Thermolyse von alpha-Azidozimtestern führt über die 2H-Azirine mit sehr guten Ausbeuten zu 2-Indolcarbonestern. Ein Mechanismus für die Enazid-Thermolyse wird diskutiert.
Thermolysis of alpha-azidocinnamtes gives, through 2H-azirines a good yield of indol-2-carboxylates. A mechanism of the enzaide thermolysis is discussed.
Step 3: Decarboxylation of indole-2-carboxylates
J. Chem. Soc., 1956, 4589-4592 (../rhodium/pdf /tryptamines.
(i) 5-Methoxytryptamine-2-carboxylic acid (2 g.) was boiled under reflux with 5% hydrochloric aicd (30 c.c.) for 1 hr. The solid soon dissolved with evolution of carbon dioxide. The cooled solution was made alkaline with 30% aqueous sodium hydroxide, the colourless oil was extracted with ether, and the ether dried (MgSO4) and evaporated, giving 5-methoxytryptamine (1.45 g).
(ii) The amino-acid (0.5 g) and toluene-p-sulphonic aicd (4.2 g) in water (5 c.c.; 5N-solution) were boiled under reflux for 1 hr. The solution was made alkaline and extracted with ether, giving 5-methoxytryptamine (0.2 g).
J. Braz. Chem. Soc., 2003, 14(1), 11-15 (http://jbcs.sbq.org.br/jbcs/2003/v14_n1
A mixture of methyl-5,6-methylenedioxyindol-2-ylcarbo
A mixture of 5,6-methylenedioxyindol-2-yl-methanoic acid (8) (0.72 g, 3.5 mmol) and barium hydroxide (0.17 g, 0.55 mmol) was finely ground and heated in “cold trap” using a Bunsen flame under vacuum (20-30 mmHg). The solid was sublimed to provide the pure indole (0.47 g, 84%).
Synthesis of Methyl azidoacetate
J. Braz. Chem. Soc., 2003, 14(1), 11-15 (http://jbcs.sbq.org.br/jbcs/2003/v14_n1
A mixture of sodium azide (26 g, 400 mmol) in water (24 mL) was added by stirring to a solution of methyl bromoacetate (50 g, 327 mmol) in methanol (50 mL). The resulting mixture was refluxed for 4 h after which it was cooled to 25 °C and the methanol removed under reduced pressure. The crude product was purified under reduced pressure distillation (bp 72-76 °C, 30 mmHg), to provide the methyl azido acetate as a clear liquid (32.3 g, 99% yield).
There are dozens of decarboxylation method, most with Cu/quinoline, but also with microwaves. It seems as if the decarboxylation of the 2-position is much easier than with tryptophan.
Enazide, 1. Mitt.: Synthese von alpha-Azidozimtsäureestern
Enazides, I: Synthesis of Ethyl alpha-Azidocinnamates
H. Hemetsberger, D. Knittel und H. Weidmann
Monatshefte, 1969, 100, 1599-1603

Abstract: Durch Kondensation von Azidoessigester mit verschieden substituierten Benzaldehyden wurden eine Reihe von alpha-Azidozimtsäureestern erhalten und deren UV- und IR-Spektren aufgenommen. Die Konfiguration dieser Verbindungsklasse ergibt sich aus NMR-Untersuchungen.
A series of alpha-azidocinnamic esters have been prepared by condensation of ethyl alpha-azidoacetate with various substituted benzaldehydes; their IR and UV spectra have been recorded. Conformations of the compound were assigned on the basis of NMR spectra.
Enazide, 2. Mitt.: Synthese von 1,3-Diaryl-2-azido-propenonen (alpha-Azidochalkonen)
Synthesis of 1,3-diaryl-2-azido-propenones (alpha-Azido-chalcones)
D. Knittel, H. Hemetsberger, H. Weidmann
Monatshefte, 1970, 101, 157-160

Abstract: Durch Kondensation von verschiedenen omega-Azidoacetophenonen mit aromatischen Aldehyden wurde eine Reihe von alpha-Azidochalkonen erhalten.
Some alpha-azido-chalcones are prepared by condensation of omega-azidoacetophenones with aromatic aldehydes.
Enazide, 4. Mitt.: Synthese und Thermolyse von alpha-Azidoacrylestern
Enazides, IV: Synthesis and Thermolysis of alpha-Azidoacrylates
H. Hemetsberger, D. Knittel
Monatshefte, 1972, 103, 194-204

Abstract: Durch Kondensation verschiedener aromatischer bzw. heterocyclischer Aldehyde mit Azidoessigester oder omega-Azidoacetophenon wurden substituierte Vinylazide hergestellt, deren Thermoylse zu kondensierten Pyrrolen führt. Der Mechanismus wird diskutiert.
By condensation of various aromatic and heterocyclic aldehydes with ethyl azidoacetate or omega-azidoacetophenones substituted vinyl azides were prepared, which yield condensed pyrroles on thermolysis. The mechanism is discussed.
The tendency is to push it as far as you can
(Hive Adickt)
09-18-04 12:23
No 531967
Here are some other ref's. Post 262291 (hest: "Re: 4-acetoxy-DiPT - newbee talking out arse, here", Tryptamine Chemistry)
The first step from a 2-bromo-accetic ester is easye to do. And the azide is stabel for years. I'm sure that any ester will work.